
Protocol Amendment Translation: A Short Guide
Protocol amendment translation is a challenging but key service in the life sciences translation field. Amendments are changes or modifications made to documents, specifically, clinical

Protocol amendment translation is a challenging but key service in the life sciences translation field. Amendments are changes or modifications made to documents, specifically, clinical

Life Sciences is a diverse and interdisciplinary field that encompasses a wide range of scientific disciplines; all of them focus on understanding complex living organisms

Translating Informed Consent Forms (ICFs) usually brings up a debate, because sometimes the barrier to understand is not just which language the ICF is in,

Open Enrollment is an annual period when United States citizens can sign up for new health insurance, modify their current plan, or cancel it altogether.
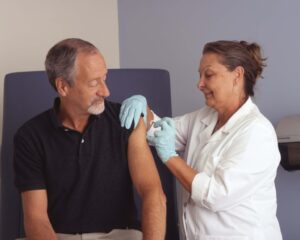
Medical translation plays a major role in bridging communication gaps between healthcare providers and their multilingual patients. In the United States, medical organizations must find

As its name suggests, linguistic validation is a process used to ensure the translated content of Clinical outcome assessments (COAs), and especially patient-reported outcomes (PROs),

The European Union (EU) implemented new regulations concerning medical and in vitro devices called: the MDR & IVDR regulations. One of the important points that

Since 2017, the European Union has been designing a series of new regulations directed to improve the safety of medical devices (MDRs) and in vitro

Accessing health information is a right, not a privilege. Health Literacy Month is a grassroots campaign aimed at promoting the importance of understandable health communication.

Multilingual solutions play a fundamental role in clinical trial development, in particular when it comes to patient recruitment, retention, and follow-up. In this blog, we